Today was our first day back from break, and before we left for break we had believed to be completely done with the basic body of our rocket. However, we soon discovered that there were some gaps in the body, so we filled the smaller ones with super glue. After we had finished that, we saw a considerable gap that could definitely not be fixed with super glue. We asked Andrew for suggestions
|
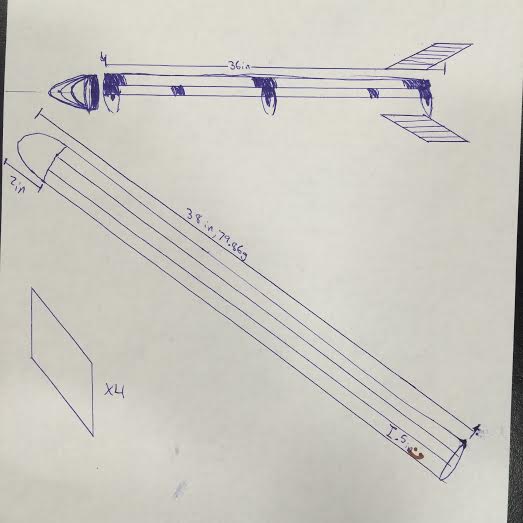
Today was the day we finalized our rocket design. Garrett suggested that we should make the rocket out of balsa wood to minimize the amount of weight, and I thought it was a good idea as well. In this design, we would get about 6 dowels and protecting them would be 8 thin pieces of balsa wood, acting as a tube. The fins we weren't to sure how to make, but as I searched online I figured the best shape would probably be a parallelogram, as supposedly that was the best for aerodynamic flow. Our parachute would be attached to our nose, so we would pop the nose of our cone and with that the parachute would come out.
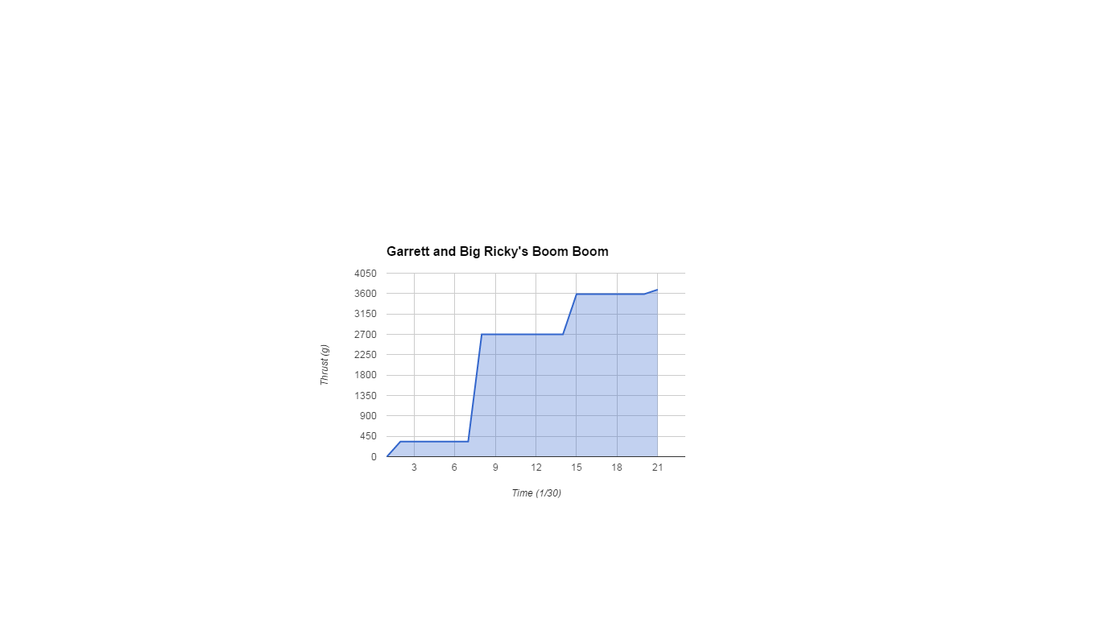
Today was our chance to see which motor we wanted to choose to integrate into our final rocket. Since Garrett and I's motor CATO'd, we had to choose another motor to use. Garrett and I decided to use Philip and Gabe's motor, as they had a greater amount of total thrust than pretty much any of the other motors.
Today we got our motor test video back and we were to figure out our thrust curve with it. To see that, first we had to put the video in an editing program so that we could see the amount of thrust occurring every fraction of a second. Once we figured all the numbers for all the fractions of seconds, we put the information into google spreadsheets and made our graph.
Today, we had a lab experiment in where we had a little introduction to earth sciences. In this intro, we would learn about what the Earth was made of and make our own chemical reactions as well. To start off, Andrew told us about what the Earth was made of. He explained that in the very middle was the core, which was made of solid iron. Next, came the outer core, made of liquid iron. Following the outer core was the mantle, made of molten rocks and last was the crust, made from solidified mantle.
Next, we took a closer look at the table of elements and he told us how we can find out whether or not two elements will have a chemical reaction or not. The way we find this out is called electronecativity. He explained that this was used to describe how easy it is for the electron to take away from the right side. To explain this, he drew a line through the table of elements, going up diagonally to the right. He said that the further elements are from each other in accordance to the table of elements, the more they react. After, we got on to the lab portion of this lesson. He told us that we were going to put some substances in vials of sodium silicate and calcium chloride. He explained to us that these substances wouldn't dissolve in the vials because they were aqueous, meaning that they couldn't dissolve in water. Next, he put 4 different substances in 4 vials. We found that the first one formed a white crystal of a sort, and the next two seemed to be the same substance, and formed pretty quickly as with the first one. However, by far the longest was the last one, which resulted in a blue substance. We found that all of the substances dragged down at first, then expanded to the top. However, the substances that expanded the most were the blue one and the two identical ones that were purple/blue in color. Overall, it was an alright lab experiment. I felt that the combustion one was cooler, but this one was pretty interesting as well. What caught my interest was the talk of chemical reactions and actually seeing the substances form into crystals over time. I feel we did this lab to get more of a feel for what's around us in this planet, and to get to know what the planet itself is actually made of. Also, this served to tell us a bit more about how chemical reactions work, which I feel will prove to come in handy in the future as this is a chemistry class. Today we got to test the motors that we had designed the first week of this section. Since Brian Delgado's class were also designing rocket motors, we decided to partner up with their class and do the tests all together. For this, we headed over to NTC park. We had to stand about 50 yards away as a precaution, so that we could eliminate any risk of a motor hitting us. After a few motors, the motor I had designed with Garrett came up. When we launched our motor, it sadly didn't take off in the sky as we had hoped. Instead, it flew straight off the stand on to the ground.
|
Ricky's Chemistry BlogHere you will find all about the experiments we are doing in chemistry as well as the individual progress I am making. Archives
December 2015
Categories |
 RSS Feed
RSS Feed
